
When p&T is unchanged, then gas volume has a direct relationship with the number of electric moles. When p & n is constant, the gas volume carries a direct relationship with temperature. Therefore, atmospheric pressure must be added to gauge pressures to convert them to an absolute basis. (2) For economic reasons, most (but not all) pressure gauges read zero pressure when pressure is equal to the ambient atmospheric pressure. The entropy can be regarded as a proportionality constant between the change in free. The universal gas constant in practical units is. In a constant n&T, the volume carries the opposite relationship to the pressure generated by the gas. The gas constant is equal to Avogadro's constant times Boltzmann's constant, the latter serving as a proportionality constant between the average thermal (kinetic) energy of the particles in an ideal gas and the temperature: ( U T) p 3 2 k B. Let's look at the pressure in this gas as ‘p,’ Note: From the SI system the amount of constant gas is 8.314 kJ / mol.K

The pressure times the volume of a gas divided by the number of moles, moles, and the temperature of the gas is always equal to a constant number. As this formula does not have any gas constant, we can use it in any units.The factor “R” in the ideal gas law equation is known as the “gas constant”. The formula mentioned above can be used in describing the states of an ideal gas. The Gas constant (R) of an atmosphere under the US standard can be gives as R = 8.31432 × 10 3 Nm/kmo l − 1 K − 1 R=8.31432\times T 1 P 1 V 1 = T 2 P 2 V 2 So, gas constant is equal to work done by one mole of a gas if its temperature is increased by 1 K.Īccording to this definition, the value of gas constant is as follows R = 8.314 J per mole per Work done = pressure x increase in volume = P x∆V Work done by gas at constant pressure is given by following expression gas constant synonyms, gas constant pronunciation, gas constant translation, English dictionary definition of gas constant.

082 L atm per mole per kelvin.ģ) Work is done by a gas if it expands on receiving heat.
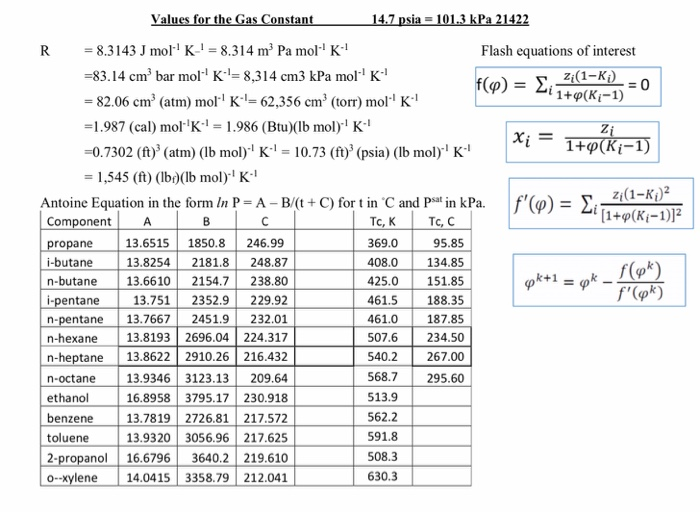
T is the temperature of the gas, measured in Kelvins. where: p is the pressure of the gas, measured in Pa V is the volume of the gas, measured in m n is the amount of substance, measured in moles R is the ideal gas constant and. It relates the energy scale to the temperature scale in. The properties of an ideal gas are all summarized in one formula of the form: pV nRT. The molar gas constant is a combination of Boyles law, Charles law, Gay-Lussac’s law and Avogadros number. It can also be written as joule per mole per Kelvin. So, When one mole of a gas is kept at temperature of 1K and pressure of one atmosphere, the volume occupied by it is called gas constant.Īccording to this definition, the value of gas constant is as follows R =. The SI unit of the ideal gas constant is Pascal or Newton per metre. How different from the characteristic gas constant and universal gas constant The universal gas constant is R 0,082057338(47) L.atm/mol.K.The specific gas constant is the ratio universal gas constant/molar mass. The situation is really no different, say, than the ideal gas law, in which the gas constant is numerically different and has different units depending on the. Equilibrium constants for several systems are listed in Table 18-2. That is why gas constant is also known as universal gas constant.Ģ) If P = 1, T = 1, V = R. Gas Constant in Different Unit Systems The adsorption equilibrium constants Ka c a.c will be in different units for liquid systems than for gas systems. 11.10 Useful Constants, Definitions, and Conversion Factors Universal gas constant R u 8.31451 J / mol K 1.98589 Btu / mol R Heat transfer rate W / m 2 8.806 × 1 0 5 Btu / ft 2 s Heat of vaporization Lv: The quantity of heat required to convert a unit of liquid at a specific temperature into its vapor at the same temperature. dure should be identical to the gas permeability constant obtained by the gas phase experiment, since the true driving force is the partial pressure differ- ence of the gas, which is independent of the concentration of 02 in the phases involved (i.e., gas phase or dissolved gas). In Ideal Gas Law, theres gas constant R that we need to use in the formula PVnRT. This value is applicable for any ideal gas. The unit for the gas constant is the joule per mol-kelvin. Gas constant can be defined in many ways, some of which are given below.ġ) If V be the volume of one mole of a gas at absolute temperature T and pressure P then the ratio of, product of P and V, and value of T is called gas constant which is represented by R, so, The currently accepted value for the universal gas constant R is: R Constant 8.3144598 J/molK.
